ConforMIS Launches Next Generation Patient-Specific Total Knee
iTotal® platform designed with specific features to address potential causes of patient dissatisfaction
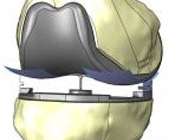
BURLINGTON, Mass.–(BUSINESS WIRE)–ConforMIS, Inc. today announced the commercial availability of its iTotal® G2 Knee Replacement System, the only patient-specific total knee replacement (TKR) available on the market today. The iTotal G2 is the next generation version of the iTotal which was FDA cleared in January 2011 and launched into limited surgeon release in May 2011. The iTotal G2 incorporates improvements, based on a year’s worth of surgeon feedback, to the implants, instrumentation and patient specific planning guides.
Studies have shown that 1 in 5 patients with a total knee replacement system are not satisfied with the results of their surgery. Issues such as implant overhang, errors in component placement and off-the-shelf implant shapes that do not match the patient’s native anatomy can result in residual pain, functional limitations and a knee that does not feel natural. By preserving the core benefits of the iTotal TKR system, including an anatomic implant design with personalized fit, a patient-matched anatomic shape and greater bone preservation than traditional total knee replacements, the iTotal G2 is designed to address the drivers of patient dissatisfaction.
Read the Full Article on BusinessWire